- C-MORE Home
- What is Microbial Oceanography?
- What is C-MORE?
- Research
- • Research Cruises
- » BioLINCS
- Home
- Cruise Participants
- Instruments Used
on the Cruise - Nitrogen Cycling
- Marine Microbes
- Cruise Blog
- Data Archive
- Education & Outreach
- People
- Publications
- Image Library
- Contact Us
Nitrogen Cycling in the Open Ocean
Nitrogen is one of the most important elements for biological systems. While we think of air as containing oxygen and maybe a little carbon dioxide, it’s mostly (80 %) nitrogen. Nitrogen is also one of the basic elements of life, along with carbon, hydrogen, phosphorus, and oxygen.
Although nitrogen is incredibly abundant in the air we breathe, it is often a limiting nutrient for the growth of living organisms. This is because the particular form of nitrogen found in air—nitrogen gas—cannot be assimilated by most organisms. Most plants, algae, and microbes require biologically available, or “fixed” forms of nitrogen, such as ammonia or ammonium. That’s why these compounds are a primary component of many plant fertilizers.
In the open ocean, as on land, fixed nitrogen is one of the most important growth-limiting nutrients for photosynthetic organisms (primary producers) such as algae and marine bacteria. Nitrogen can also serve as an energy source or as an oxidant for marine bacteria and archaea. (Archaea are single celled organisms that look similar to bacteria, but which are in an entirely separate biological domain).
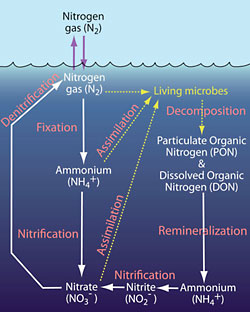 The ocean absorbs nitrogen gas from the atmosphere. In open-ocean areas with low concentrations of nutrients (“oligotrophic” regions), some of this nitrogen is taken up by microbes and transformed into various chemical compounds. The key steps in this process are listed below and shown in the simplified diagram at right (click on it to see a larger version).
The ocean absorbs nitrogen gas from the atmosphere. In open-ocean areas with low concentrations of nutrients (“oligotrophic” regions), some of this nitrogen is taken up by microbes and transformed into various chemical compounds. The key steps in this process are listed below and shown in the simplified diagram at right (click on it to see a larger version).
- Nitrogen gas (N2) from the atmosphere dissolves into seawater at the ocean surface. Nitrogen gas is the most abundant form of nitrogen in the ocean, but is not useful to most living things.
- Dissolved nitrogen gas is taken up by just a few types microbes, which convert the nitrogen into a much more useable form, known as ammonium (NH4+). This process, known as “nitrogen fixation,” is vitally important. Without it, very little nitrogen would available for thousands of other organisms that live near the ocean surface.
- Ammonium is the form of nitrogen that is most easily consumed by microorganisms. For this reason, ammonium is consumed almost as fast as it is produced, a process called “assimilation.” The result is that the nitrogen becomes incorporated into the cells of living organisms.
- Some marine microbes consume nitrite and nitrate, another form of assimilation.
- When microbes (and other organisms) die, they decompose, releasing ammonium and tiny particles containing particulate organic nitrogen (PON), as well as dissolved organic nitrogen (DON) into the surrounding seawater.
- Some microbes convert ammonium to nitrite (NO2-) and then nitrite to nitrate (NO3-). This two-step process is called “nitrification.” The result of this process is that nitrate is released into the ocean.
- A host of organisms consume particulate organic nitrogen and dissolved organic nitrogen, converting some of the nitrogen back to ammonium. This process is called “remineralization.”
- To complete this complex cycle (which begins to look more like a Celtic knot), some microbes convert nitrate and nitrite back to nitrogen gas through a process called “denitrification.”
The open ocean is host to a wide variety of microbes, each adapted to a perform one or more steps in the nitrogen cycle. This enormous diversity of microbes has evolved to use every form of nitrogen in the ocean, either as a source of energy or as a nutrient. However, “reduced” nitrogen compounds, such as ammonium are easier for most microbes to use than “oxidized” nitrogen compounds such as nitrate.
Many types of microbes extract energy from nitrogen compounds by “oxidizing” these compounds (for example, converting ammonium to nitrite or nitrite to nitrate). However, relatively few microbes can “reduce” nitrogen (for example converting nitrogen gas to ammonium), because this requires extra energy, which they must get from other sources (“food” and/or sunlight).
For more information on some the different types of nitrogen-processing microbes we’ll be studying during the BioLINCS cruise, see the marine microbes page.
[ Top of Page ]



